Psoriasis: Pathogenesis, Treatment, and Evolving Disease Models

Introduction:
Autoimmune skin diseases (AISDs) are a group of chronic inflammatory disorders characterized by the immune system mistakenly recognizing self-antigens of the skin as foreign, leading to autoantibody production, immune cell activation, and subsequent skin tissue damage. Common AISDs include psoriasis vulgaris (psoriasis), atopic dermatitis (AD), vitiligo, and bullous pemphigoid (BP), among others [1, 2].
Among these, psoriasis stands out for its high global prevalence, affecting 2–3% of the population worldwide. It is a chronic, relapsing-remitting disease that poses significant physical and psychological burdens on patients [2]. Current therapeutic strategies primarily focus on symptom management, including topical agents, phototherapy, and systemic treatments. Recent breakthroughs in biologics, small-molecule drugs, and targeted therapies have revolutionized psoriasis treatment, shifting the paradigm from symptom control to disease modification. This article discusses the pathogenesis, clinical impact, and evolving treatment landscape of AISDs, with a particular focus on psoriasis.
Pathogenesis of psoriasis
Psoriasis is a chronic immune-mediated skin disorder triggered by the interplay between genetic susceptibility and environmental factors (e.g., infections, injury, or medications). Based on clinical phenotypes and progression, psoriasis can be classified into several subtypes, including plaque psoriasis (psoriasis vulgaris), guttate psoriasis (scattered, drop-like lesions), erythrodermic psoriasis, pustular psoriasis, and psoriatic arthritis.
The disease primarily stems from abnormal keratinocyte hyperproliferation and dysregulated differentiation, accompanied by impaired skin barrier function. These dysfunctions activate innate immune cells, such as dendritic cells (DCs) and macrophages, which secrete pro-inflammatory cytokines like interleukin-23 (IL-23) and tumor necrosis factor-α (TNF-α). These cytokines drive the activation and maintenance of the T helper 17 (Th17) cell pathway, further stimulating keratinocytes to produce more pro-inflammatory cytokines and chemokines, which leads to hallmark pathological and clinical features, including epidermal thickening (acanthosis), scale formation, and dermal vascular dilation (Figure 1) [3].
Figure 1. Pathogenesis of psoriasis [3]
Current clinical landscape of psoriasis therapeutics
The treatment of psoriasis is tailored based on disease subtype, effected body surface area, and severity:
- Localized plaque psoriasis: topical therapies such as corticosteroids, vitamin D analogs (e.g., calcipotriol), or tazarotene are first-line options.
- Extensive lesions: phototherapy (e.g., UVB) is effective but carries long-term carcinogenic risks.
- Severe or joint-involved psoriasis: systemic agents are employed, including methotrexate, immunosuppressants (hydroxyurea, 6-mercaptopurine, azathioprine), and the PDE4 inhibitor apremilast.
Over the past two decades, targeted biologics against pro-inflammatory cytokines (e.g., anti-TNF, anti-IL-12/23, anti-IL-17, and anti-IL-23 monoclonal antibodies) have become cornerstone therapies for moderate-to-severe psoriasis due to their high efficacy and acceptable safety profiles [4].
Recent years have witnessed accelerated drug development, with novel oral small molecules and next-generation biologics entering clinical use or trials (Table 1) [4]. The overall trend is coexistence of more oral and biologic agents targeting key signaling pathways, balancing efficacy, safety, and dosing convenience [4]:
- Deucravacitinib (Bristol-Myers Squibb): an FDA-approved allosteric TYK2 inhibitor, demonstrating superior short- and long-term efficacy over placebo and apremilast.
- Bimekizumab: a dual anti-IL-17A/F monoclonal antibody achieving exceptional response rates in Phase III trials.
- Pipeline candidates: oral IL-17 inhibitors (DC-806, DC-853), next-gen PDE4 inhibitors (orismilast, ME3183), oral IL-23 receptor antagonist (JNJ-2113), dual TYK2/JAK1 inhibitors
Table 1. Selected approved and investigational psoriasis therapies [4]
Novel drug development strategies for psoriasis
Despite remarkable therapeutic advancements, critical unmet needs persist in clinical practice [5], including:
- Mitigating drug-related adverse effects: Current immunosuppressants may increase risks of infections, cardiovascular abnormalities, and even malignancies.
- Improving long-term efficacy maintenance: Reducing relapse after discontinuation and minimizing the burden of sustained therapy.
- Achieving higher therapeutic benchmarks: Pursuing more complete skin clearance and developing targeted therapies for refractory cases or difficult-to-treat areas.
To address these challenges, diversified R&D strategies are emerging. While optimization of small-molecule drugs and antibody-based biologics continues, novel approaches are being explored, such as peptide, cell-based therapies, and microbiome-modulating treatments. Among these, preclinical in vitro assays and in vivo animal models play a vital role in the development of psoriasis drugs:
- Cell cultures and co-culture models elucidate drug effects on inflammatory pathways, keratinocyte proliferation/differentiation, and enable rapid candidate screening.
- Evaluate efficacy in complex skin microenvironments, compare delivery routes/formulations, and assess preliminary pharmacokinetics/toxicology.
- Identify translational biomarkers to de-risk clinical trials and reduce development costs.
The Immunology Center in WuXi Biology offers a comprehensive suite of preclinical services to accelerate drug development for autoimmune and inflammatory diseases, including psoriasis, asthma, and systemic lupus erythematosus (SLE). Our integrated in vitro and in vivo platforms provide critical support for novel therapeutic discovery in AISDs (Figure 2).
Figure 2. WuXi Biology empowers drug discovery for autoimmune and inflammatory diseases
Case study 1: in vitro Th17 functional assays and imiquimod (IMQ)-induced Psoriasis Rat models
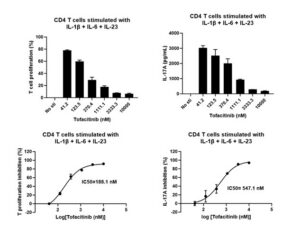
In psoriasis pathogenesis, CD4⁺ T-cell imbalance (particularly Th17 hyperactivation) is a key driver of chronic inflammation. To evaluate drug candidates targeting the pathogenic Th17/IL-23 axis, WuXi Biology developed an in vitro Th17 functional assay. Using purified human CD4+ T cells stimulated with a cytokine cocktail, we demonstrated that the pan-JAK inhibitor tofacitinib significantly suppressed T-cell proliferation and IL-17A secretion, validating JAK/STAT pathway inhibition as a therapeutic mechanism (Figure 3).
Figure 3. In vitro activation and inhibition assays of Th17
Imiquimod is a TLR7 agonist that promotes DC secretion of IL-23 by activating TLR7, thereby activating the Th17 immune response pathway. WuXi Biology successfully induced skin lesions resembling human psoriasis by applying imiquimod cream to the back of rats. The severity of psoriasis-like inflammation was assessed through clinical dermatitis scores. Results showed that the skin of the control group remained intact, whereas the IMQ-induced group exhibited typical psoriasis-like symptoms, including pathological features such as skin erythema, scaling, and thickening. After treatment with the pan-JAK inhibitor tofacitinib, these symptoms were significantly alleviated (Figure 4A). Histopathological analysis further confirmed that the skin tissues of the IMQ-induced model group displayed characteristic psoriatic changes, including marked inflammatory cell infiltration and epidermal thickening (Figure 4B).
Figure 4. IMQ-induced psoriatic skin lesions in rats
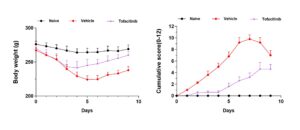
Simultaneously, it was observed that the skin of the control group remained normal, whereas the model group showed a significant increase in clinical PASI scores. After tofacitinib intervention, the scores decreased markedly. The distinct differences in clinical scores among the three groups indicate the successful establishment of this model (Figure 5). Additionally, the therapeutic window observed between the IMQ-induced group and the treatment group suggests that this model can be used for evaluating the efficacy of anti-psoriatic drugs.
Figure 5. Body weight and clinical scores in IMQ-induced psoriatic rats
Case study 2: In vitro IL-23/IL-17 cell activation assays and in vivo humanized mouse models
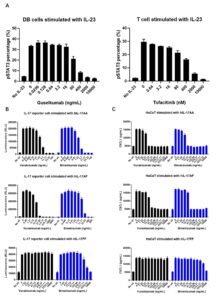
In psoriasis, the IL-23/Th17/IL-17 axis is a key pathogenic pathway. IL-23 primarily promotes the differentiation and proliferation of Th17 cells, which secrete pro-inflammatory cytokines including IL-17. Upon binding to its receptor (IL-17R) on keratinocytes, IL-17 drives abnormal keratinocyte differentiation and proliferation, as well as the expression of chemokines (CXCL1, CXCL2, and CCL20), thereby amplifying skin inflammation. To evaluate the regulatory effects of candidate drugs on this signaling pathway, WuXi Biology established in vitro cell activation assays for IL-23/IL-17. The effects of drugs on the IL-23 pathway were assessed by detecting pSTAT3 in cells stimulated with IL-23 (Figure 6A). Additionally, a reporter gene cell line (Figure 6B) was used to rapidly screen drugs, such as antibodies that block the binding of IL-17 and IL-17R. Furthermore, the functional effects of the drugs on the IL-17 signaling pathway were validated using IL-17 activation experiments in keratinocytes (Figure 6C), which assessed pro-inflammatory factors secreted by the keratinocytes.
Figure 6. In vitro functional assays of IL-23 and IL-17 signaling pathways
WuXi Biology successfully induced skin lesions, resembling human psoriasis, in a B-hTL1A/hIL23A/hIL12B humanized mouse model using localized application of IMQ. The experimental results showed that the skin of control (sham) mice remained intact and free of erythema and scaling. In the vehicle group, mice displayed significant erythema, thick scales, and skin thickening starting on day 3, with the total PASI score increasing significantly by day 7, markedly higher than that of the sham group. In mice treated with the anti-IL-23 monoclonal antibody, guselkumab, the skin lesions rapidly relieved, and the PASI scores were significantly lower compared to the vehicle group (Figure 7).
Figure 7. Clinical score and skin lesions in IMQ-induced psoriatic mice
At the same time, H&E staining revealed that the vehicle group exhibited marked epidermal acanthosis, parakeratosis, and dense inflammatory cell infiltration. In contrast, the guselkumab-treated group showed significant improvement, with notably reduced inflammatory infiltration. These findings demonstrate that the team successfully established a psoriasis model in B-hTL1A/hIL-23A/hIL-12B humanized mice using IMQ induction. This model is responsive to IL-23-targeted therapy, making it suitable for preclinical evaluation of novel anti-psoriatic drugs (Figure 8).
Figure 8. H&E staining of IMQ-induced psoriatic mice
Summary
Beyond the aforementioned models, the Immunology Center in WuXi Biology has established a series of in vitro experimental assays to investigate the pathogenesis of psoriasis, drug interactions and drug safety assessment. Key assays include:
- CD4+ T cell and Th17 cell induction, differentiation, and functional assays;
- The release of pro-inflammatory cytokines in keratinocytes;
- IL-17, IL-36, and TNF-α signaling pathway studies (Figure 9A).
Additionally, WuXi Biology has developed well-characterized in vivo models in mice, rats, and non-human primates (NHPs). These models support comprehensive pharmacodynamic, pharmacokinetic, and biomarker studies for psoriasis drug development (Figure 9B).
Figure 9. WuXi Biology In vitro assays and in vivo animal models for psoriasis research
References
- Zhang B, Mei X, et al. J Transl Autoimmun. 2024 Jan 12;8:100232. doi: 10.1016/j.jtauto.2024.100232.
- Lai et al. Clinical Epigenetics (2025) 17:182, https://doi.org/10.1186/s13148-025-01999-6
- Greb, J., Goldminz, A., Elder, J. et al. Nat Rev Dis Primers 2, 16082 (2016). https://doi.org/10.1038/nrdp.2016.82
- Al-Horani R, Chui T, Hamad B. 2024 Jul;23(7):492-493. doi: 10.1038/d41573-024-00018-2.
- Guo, J., Zhang, H., Lin, W. Sig Transduct Target Ther 8, 437 (2023). https://doi.org/10.1038/s41392-023-01655-6
Related Content
Inflammatory bowel disease (IBD), which includes ulcerative colitis and Crohn’s disease, is a chronic, immune-mediated disorder characterized by persistent inflammation...
VIEW RESOURCEInflammatory bowel disease (IBD), which includes ulcerative colitis and Crohn’s disease, is a chronic, immune-mediated disorder characterized by persistent inflammation...
VIEW RESOURCE