How to Accelerate the Synthesis of PROTACs: Strategies and Case Studies
Introduction:
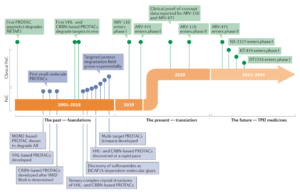
Targeted Protein Degradation (TPD) has emerged as a promising therapeutic modality due to its potential to develop therapeutics for previously undruggable targets and address the resistance issues of small molecule inhibitors. Central to this strategy is the novel protein-degradation approach represented by proteolysis-targeting-chimeras (PROTACs), which induce targeted protein degradation through the ubiquitin-proteasome system (UPS). Research interest has rapidly shifted towards the PROTAC modality with the entry of Arvinas’ PROTACs, ARV-110 and ARV-471, into Phase I clinical trials (Figure 1). Subsequently, PROTAC molecules from companies like Nurix Therapeutics and Kymera Therapeutics have advanced into clinical stages. Encouraging clinical results further endorse the PROTAC concept and validate the safety and efficacy of such molecules, inspiring the next generation of therapeutic tools.
Figure 1. Timeline of PROTAC discoveries
(Source: Craig M. Crews et al. Nat. Rev. Drug. Disc. 2022, 21, 181)
Although categorized as small molecules, PROTACs exceed the boundaries established by Lipinski’s “Rule of Five” (Ro5) for optimal physicochemical properties, especially with regard to molecular mass and the number of hydrogen bond donors and acceptors. This deviation poses significant challenges in the synthesis and development of potential drug candidates. The core and technical complexity of PROTAC drug discovery and development rest in the swift and efficient synthesis of PROTAC molecules. Surmounting this hurdle generally necessitates the synthesis of an extensive series of molecules for thorough investigation of structure-activity relationship (SAR).
Synthesis Strategies for PROTACs
PROTACs are heterobifunctional molecules consisting of: (1) a ligand that binds to a protein of interest (POI); (2) a ligand for recruiting an E3 ubiquitin ligase to promote POI ubiquitination; and (3) a linker connecting these ligands. From a chemical synthesis perspective, PROTACs are built around these three core components. Each component can be linked through easily formed chemical bonds, a feature that facilitates the modular synthesis of PROTACs.
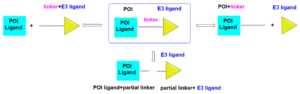
At present, three main assembly methods are utilized (Figure 2): (1) connecting the E3 ligase ligand to the linker, then attaching the POI ligand; (2) initially joining the POI ligand to the linker, followed by coupling with the E3 ligase ligand; and (3) securing a section of the linker to both ligands before assembling them together. Given their large molecular weight and multiple functional groups, PROTACs necessitate relatively mild conditions in later synthetic stages to minimize the risk of side reactions. Common chemical reactions in PROTAC synthesis encompass acid-amine coupling, reductive amination, and click reactions, among others.
Figure 2. Assembly methods for PROTACs
Common E3 ligands include Von Hippel-Lindau (VHL), Cereblon (CRBN), and Inhibitor of Apoptosis Protein (IAP), as depicted in Figure 3. Frequently used linker structures are shown in the bottom panel of Figure 3. Approximately 65% of reported linkers are PEG or analogous saturated carbon chain structures. Owing to PEG’s remarkable flexibility, this moiety can facilitate the assembly of ternary complexes. As studies on protein degradation expand and PROTACs with rigid linkers progress into clinical trials, the selection process of PROTAC linkers should also include consideration of their length. The length of linkers substantially influences a PROTAC’s activity, membrane permeability, and conformation. A too short chain increases steric hindrance, preventing the formation of ternary complexes; if a linker is too long, molecular weight and flexibility rise, affecting membrane permeability and bioavailability. Currently, widely used rigid linkers include structures such as alkynes, nitrogen-containing heterocycles, piperazines, pyridines, and triazoles.
Figure 3. Common E3 ligands and linker structures in PROTACs
The synthesis of PROTACs is a complex process that requires mild reaction conditions and careful consideration of the compatibility and stability of various functional groups. The existence of numerous chiral centers in specific molecules necessitates a comprehensive understanding of chirality construction and preservation, which poses a substantial challenge. The scenario further intensifies with rigid linkers containing cis-trans isomers, as these structural attributes add intricacy to synthesis and separation procedures. The hefty molecular weight of PROTACs can induce bond breakages under vigorous reaction conditions, with CRBN ligands being particularly vulnerable to assaults by nucleophilic agents or potential ring-opening during aqueous phase quenching. Moreover, the amphiphilic characteristic of PROTAC molecules may lead them to form micelles during upscaling, potentially causing stirring difficulties and incomplete reactions, thus adding yet another level of complexity to the synthesis process.
Most PROTAC molecules currently in clinical trials utilize Cereblon (CRBN) and its analogs as E3 ligands. This is primarily due to the smaller molecular weight, fewer hydrogen bond donors and acceptors, and higher lipophilicity of CRBN ligands, which closely align with the Ro5 principles for orally administered drugs. Both Pomalidomide and Lenalidomide ligands have a single chiral center. However, given their propensity to racemize in the body, most reported compounds are racemates. As depicted in Figure 4, Compound 1 can attach the linker to positions 3, 4, 5, and 6 of the phenyl ring structure. For oxygen-linked CRBN, the standard strategy involves replacing the corresponding fluorine atom through aromatic nucleophilic substitution or forming an ether bond via the Mitsunobu reaction. Considering the acidity of the nitrogen-hydrogen on the amide and the phenolic hydroxyl group, the Mitsunobu reaction can preferentially occur on the phenolic hydroxyl group under suitable conditions, creating an ether bond. For nitrogen-containing derivatives, nucleophilic substitution, alkylation, and Buchwald reactions are typically employed. For C-C bond coupling, reactions such as Heck, Suzuki, and Sonogashira are generally used.
Figure 4. Synthetic strategies for CRBN ligands
Challenges in PROTAC Synthesis
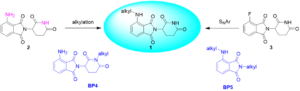
What exactly makes PROTAC synthesis so challenging, given that the coupling reactions employed are common? As illustrated in Figure 5, the synthesis of pomalidomide CRBN ligand 1 with an amino substitute involves two theoretically feasible paths: alkylation from aniline compound 2, and a nucleophilic substitution reaction originating from fluorinated compound 3. Yet, these seemingly straightforward reactions prove difficult to execute effectively. In the alkylation route, the nitrogen-hydrogen of the amide’s acidity can readily trigger a substitution reaction at this site, leading to the unintended formation of by-product BP4. Additionally, the nucleophilic substitution reaction between compound 3 and the aryl compound of a primary amine yields less than expected, and the unwanted by-product BP5 is frequently produced.
Figure 5. Challenges in the synthesis of CRBN ligands
Moreover, certain CRBN ligands are beyond the reach of standard nucleophilic substitution reactions. Although the Buchwald reaction is a common method for forming carbon-nitrogen bonds, the complexity of these molecules often necessitates a thorough screening of numerous reaction conditions and catalysts to identify the most suitable ones. It is crucial to balance both the effectiveness and cost of the palladium catalyst used in the reaction.
Case Studies in PROTAC Synthesis
WuXi AppTec Research Chemistry Services (RCS) provides integrated chemical services, covering all synthesis, analytical, and purification stages from target to hits, leads, and preclinical candidates. To cater to specific customer requirements, RCS offers a variety of service models.
RCS has synthesized more than 90,000 PROTAC molecules, including more than 1,000 linkers, over 100 E3 ligase ligands, and more than 60 series of target protein ligands pre-coupled with various linkers. Thanks to the modular approach of PROTAC molecule synthesis, once a customer chooses a POI ligand, a series of PROTAC molecules can be synthesized swiftly and efficiently.
Case Study: Delivery of Kilogram-Level Target Compound within 29 Days through a 27-Step process
The target compound in this case was a highly polar, large molecule. The existing synthesis route, a 27-step sequence, had only been demonstrated at gram-level quantities. Further complicating the situation, the client’s subsequent testing schedule necessitated delivery within an exacting 30-day timeframe. This posed a considerable challenge, considering the conventional wisdom that each scale-up iteration should not exceed a tenfold increase. Our team was charged with executing two to three rounds of scale-up validation within a single month, while concurrently enhancing separation conditions and reducing separation and purification durations. The challenges were manifold: a prolonged synthesis pathway, a stringent timeline, and intricate separation and purification procedures.
RCS coordinated with procurement, synthesis, and logistics teams through meticulous planning and continuous communication with the client. We mobilized 40 chemists, arranged into five groups, working in shifts to ensure rapid progress of the project. The result? 181 grams of compound were successfully delivered in just 29 days, paving the way for the client’s subsequent testing to proceed as scheduled. “The WuXi AppTec team turned the impossible into reality” quoted the client.
Figure 6. Delivering 181 grams of compound in 29 days
In addition, RCS has a variety of technology platforms supporting the synthesis of PROTACs. Flow chemistry platform, with outstanding mixing and heat exchange abilities, minimizes the risks associated with high-energy and high-temperature reactions and can support PROTAC synthesis from milligram to kilogram scale. High-throughput reaction condition screening platform can perform parallel screening of multiple reaction conditions to improve efficiency and reduce time for screening, of which catalyst screening includes C-C coupling and C-Hetero coupling, usually delivering samples within two working days from screening to sample delivery. The photocatalytic reaction is usually performed at room temperature, under mild conditions, and with high compatibility with functional groups, and PROTAC plus other molecules containing multiple functional groups can also be used, which is an effective supplement to conventional methods. The RCS photoredox chemistry platform, with over 100 reactors and more than 30 catalysts, can conduct screening of more than 50 reaction types and synthesize products from milligrams to kilograms.
PROTAC Molecule Analysis and Separation
PROTAC molecules, once synthesized, often necessitate further purification to attain the required purity level for activity testing. Their large molecular weight and low solubility make separation and purification a complex process. Moreover, the multitude of functional groups in PROTAC molecules means that traditional HPLC separation could result in molecular degradation, thereby necessitating quicker and more efficient separation methods. For instance, the amide of CRBN can readily undergo ring-opening during the purification process. Furthermore, the high polarity of PROTAC molecules leads to significantly reduced retention time, necessitating a highly polar system for separation—a requirement that remains true even for SFC separation.
Addressing these challenges, the RCS analytical team has pioneered a PROTAC-specific analysis and purification platform. To date, this team has developed over 200 methodologies, achieving a remarkable 95% success rate in one-time separation.
Detection of PROTAC Ternary Complexes via Native MS
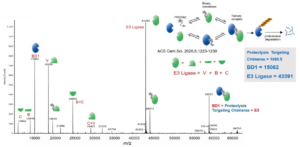
Following synthesis, the efficacy of PROTACs to bind specifically with the target protein and E3 ubiquitin ligase becomes a vital concern. Since the ternary complex is non-covalently bound, the binding strength is relatively weak and can be easily disrupted using denatured mass spectrometry methods, making the detection of the complex’s ion peak impossible. Native mass spectrometry presents a solution to this issue. It can identify the ternary complex without disrupting the non-covalent interactions within the molecules. The detection of a peak at 59557 in the mass spectrum (Figure 7) signifies the existence of the ternary complex.
Figure 7. Detection of PROTAC ternary complexes via native MS
Proteomics Study of PROTAC Off-Target Effects
Off-target effects and non-specific degradation are prevalent side effects of PROTACs, with the latter potentially leading to lethal toxicity and subsequent development failure. Early-stage candidate screening based on degradation occurrence can help avert or lessen significant losses in later research and development stages. Currently, proteomics can be employed to characterize off-target effects. The RCS analytical team has developed a proteomics platform that, when combined with Mass Spectrometry (MS), can offer insights into off-target effects.
As demonstrated in Figure 8, known drugs are utilized as samples. Different isotopes are used to label different samples, ensuring proteins within the same sample share the same reporter ions, while proteins in different samples have distinct reporter ions. Subsequently, MS is performed. The relative intensity of proteins in each sample is determined through the relative intensity of the isotope reporter ion peaks. The results, depicted as a volcano plot, reveal three noticeable degradations, all of which are anticipated. This suggests that the compound does not cause non-specific degradation. These findings were further validated using a HiBiT assay, producing similar results.
Figure 8. Proteomics research to assess the off-target effects of PROTACs
When it comes to assessing activity, the ability to determine whether a target protein, particularly those within tissue, can be degraded is crucial. The gold standard for protein identification is Mass Spectrometry (MS). In line with this, our team utilizes a sophisticated approach – Mass Spectrometry-based Targeted Proteomics methodology. This allows us to measure the relative peak intensity of target proteins in both tissue and blood with remarkable precision. Compared to conventional proteomics, Targeted Proteomics provides exceptional quantitative accuracy and a unique capability to identify proteins, even those with extremely low abundance. In addition, the method does not rely on antibody of which the quality is always a concern.
One-Stop PROTAC Platform
Beyond providing synthesis, analysis, and purification services for PROTACs, WuXi AppTec extends its support with a comprehensive suite of tests. These include drug discovery services, biological testing, DMPK, toxicology studies, and support for both CMC and commercial manufacturing. Our commitment at WuXi AppTec is to create a robust, all-encompassing PROTAC platform, thereby serving as a one-stop solution that truly empowers our customers.
Contact us: chemistry_service@wuxiapptec.com
PROTAC refers to Proteolysis Targeting Chimeras, PROTAC is the abbreviated term used for this context.
WuXi AppTec | Targeted Protein Degradation Services:
- Learn about our extensive platform of services to support TPD research by clicking HERE
- View our White Paper, entitled: “Emerging Drug Discovery Strategies for Targeted Protein Degradation” by clicking HERE
- View our Poster from DDC 2023, entitled: “Biophysical & Functional Characterization of Bifunctional Small Molecules Enables TPD Drug Discovery” by clicking HERE
Related Content
In our latest publication, WuXi AppTec scientists introduce two new methods that utilize readily accessible aldehydes to create substituted isoxazolines...
VIEW RESOURCEIndoleamine 2,3-dioxygenase 1 (IDO1) is an enzyme that metabolizes tryptophan, and its activity can lead to immunosuppression, thereby allowing tumors...
VIEW RESOURCE