Radionuclide Drug Conjugates in Anticancer Treatment

Mechanisms, Recent Progress, and Animal Models
Radionuclide drug conjugates (RDCs), a novel class of precision tumor-targeting therapeutic and diagnostic agents, consist of a targeting ligand, a linker, a chelator, and a radioactive isotope. RDCs use tumor antigen–specific molecular ligands to precisely deliver different radioactive isotopes to tumor cells, leveraging their radioactive emissions for diagnostic imaging and therapeutic effects at the target site. To date, RDCs have achieved breakthrough progress in the diagnosis and treatment of tumors such as neuroendocrine neoplasms and prostate cancer. This article explores the mechanisms of action and recent clinical advances of RDCs in cancer therapy, and provides an in-depth discussion of evaluation strategies for RDC candidates in preclinical animal models.
Mechanisms of RDCs
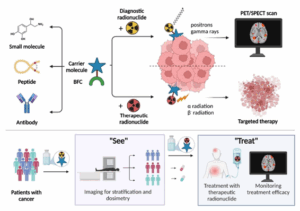
Radiotheranostics is a rapidly developing cancer treatment paradigm whose core advantage lies in coupling tumor-specific membrane protein–targeting ligands (such as peptides, small molecules, or antibodies) with radioactive isotopes, thereby combining molecular imaging and targeted therapy to achieve integrated, precision diagnosis and treatment. Depending on the functional properties of the radionuclide, RDCs can be classified as diagnostic or therapeutic.
- Diagnostic RDCs typically use radionuclides with short half-lives that emit positrons (for positron emission tomography, PET) or single photons (for single photon emission computed tomography, SPECT). These radionuclides localize targets by PET–CT or SPECT imaging and provide critical information for disease staging and assessment of treatment response.
- Therapeutic RDCs employ high-energy radionuclides that emit α or β particles. When the targeting ligand binds its target, the emitted high-energy radiation damages cellular DNA directly or increases reactive oxygen species indirectly, thereby selectively killing tumor cells (Figure 1). Examples include Novartis-developed PSMA-targeting agents 68Ga-PSMA-11 and 177Lu-vipivotide tetraxetan, used for prostate cancer imaging and therapy, respectively.
RDCs have two notable advantages over conventional targeted anticancer drugs. First, changing the radionuclide carried by the targeting ligand enables seamless integration of diagnosis and therapy. Second, whereas conventional targeted therapies act on aberrantly activated signaling pathways driven by gene mutations and can be undermined by tumor genomic instability and emergence of resistant target mutations, RDCs kill tumor cells via ionizing radiation from the radionuclide—a mechanism that does not depend on specific signaling pathways and is therefore less prone to resistance. Owing to their unique tumor-visualization capabilities, precise identification of therapeutic targets, and real-time monitoring of treatment efficacy, radiotheranostics has become one of the most precise and personalized treatment approaches to date.
Figure 1. Mechanisms of RDCs [1]
Research and clinical progress application of RDCs
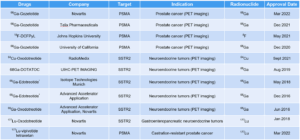
To date, 11 RDCs have been approved worldwide, of which 9 are diagnostic RDCs and 2 are therapeutic RDCs (Table 1). RDCs are currently focused primarily on cancer diagnosis and treatment. Their targets are concentrated on PSMA and somatostatin receptors (SSTR), used respectively for prostate cancer and neuroendocrine tumor management. Diagnostic RDCs predominantly employ gallium‑68 (68Ga), while all currently marketed therapeutic RDCs use lutetium‑177 (177Lu) [2].
Table 1. Global approvals of RDCs [2] (as of Dec 2023, * indicates approval by EMA)
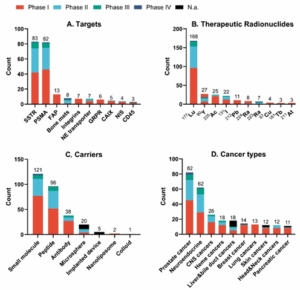
According to ClinicalTrials.gov [2], more than 270 RDC candidates are currently in clinical development worldwide. Over 100 programs are in Phase I, and 12 RDCs have advanced to Phase III (6 diagnostic and 6 therapeutic). Target distribution among investigational RDCs is highly concentrated: more than 40% focus on PSMA and SSTR2 (Figure 2A). By indication, the main target–disease pairings are PSMA (prostate cancer), SSTR2 (neuroendocrine tumors), and fibroblast activation protein (FAP) (breast, pancreatic, and lung cancers); other targets include integrin αvβ3 and HER2 (Figure 2C). Notably, 30% of new RDCs are focused on prostate cancer diagnosis and therapy (Figure 2D).
For therapeutic development, 177Lu‑based RDCs—validated by marketed 177Lu agents—are currently the first choice of therapeutic radionuclide in clinical studies, followed by yttrium‑90 (90Y), actinium‑225 (225Ac), iodine‑131 (131I), and others (Figure 2B). Diagnostic RDCs most commonly use 68Ga, followed by fluorine‑18 (18F), copper‑64 (64Cu), and similar nuclides. In recent years, α particles such as 225Ac, plumbum‑212 (212Pb), and thorium‑227 (227Th) have attracted wide attention because of their short radiation range and much higher energy (tens to hundreds of times that of β particles). These nuclides can achieve highly efficient tumor cell killing while substantially reducing damage to surrounding healthy tissue [4]. Several development programs have shifted to α particles for therapeutic RDCs; to date, more than 14 such agents have entered clinical trials.
Figure 2. Overview of RDC Clinical Trials (data as of Dec 2024) [1]
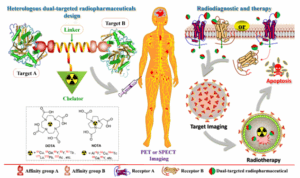
To enhance tumor targeting and therapeutic efficacy of RDCs, bispecific strategies have become a key R&D focus (Figure 3). Major target pairings currently include: PSMA with gastrin-releasing peptide receptor (GRPR); albumin (ALB) combined with targets such as folate receptor (FR), SSTR2, PSMA, FAP, and carbonic anhydrase IX (CA‑IX); integrin αvβ3 paired with targets such as FAP, SSTR2, PSMA, glucose transporters (GLUTs), GRPR, CD13, and melanocortin 1 receptor (MC1); and FAP combined with targets such as PSMA, SSTR2, biotin receptor, and GLUTs [3]. Because αvβ3 is overexpressed on tumor neovasculature and across many tumor types, it is the most frequently used partner in bispecific combinations. Bispecific RDCs involving FAP and GRPR paired with other targets have shown notable progress in indications such as breast and pancreatic cancer, with several candidates already in clinical trials. Current studies indicate that both monospecific and bispecific ligands are predominantly peptides or small molecules, with antibodies less commonly used as targeting ligands. A likely reason is that antibody-based RDCs have long biological half-lives, which can lead to nonspecific binding and increased radiation exposure to non-tumor tissues, potentially compromising safety.
Figure 3. Bispecific RDC design strategy [3]
Efficacy evaluation of RDCs in preclinical animal models
WuXi Biology evaluated the biodistribution and efficacy of RDCs in the LNCaP prostate cancer xenograft mouse model.
Case study: Diagnostic RDC — 68Ga‑RDC
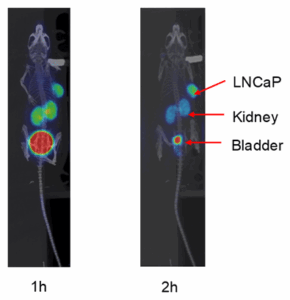
We implanted LNCaP prostate cancer cells subcutaneously into the right flank of male BALB/c nude mice. Tumor‑bearing mice were administered 68Ga‑RDC, and PET imaging showed that 68Ga‑RDC accurately identified LNCaP tumors (Figure 4).
Figure 4. PET images of 68Ga‑RDC (left: 1‑hour scan; right: 2‑hour scan)
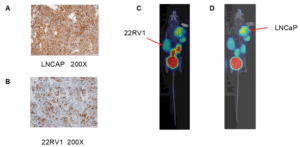
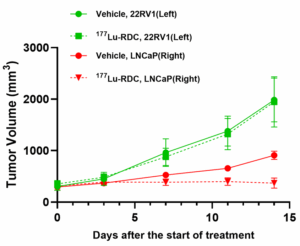
To further assess the targeting specificity of the RDC, we implanted 22RV1 cells on the left flank and LNCaP cells on the right flank of male BALB/c nude mice. After injecting 68Ga‑RDC and performing PET imaging, 68Ga‑RDC was found to accumulate markedly in the PSMA‑high LNCaP tumor but not in the PSMA‑low/medium 22RV1 tumor (Figure 5C, D). This differential distribution was consistent with PSMA expression as shown by histopathology analysis in LNCaP and 22RV1 tumors (Figure 5A, B).
Figure 5. PET imaging of 68Ga‑RDC in a dual‑tumor model. A & B. PSMA IHC staining of LNCaP and 22RV1 tumor tissues. C & D. PET images of 68Ga‑RDC in the dual‑tumor model
Case study: Therapeutic RDC — 177Lu‑RDC
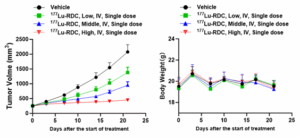
We implanted LNCaP cells subcutaneously in the right flank of male BALB/c nude mice. When tumors reached approximately 250 mm3, tumor‑bearing mice were treated with 177Lu‑RDC according to the study protocol. Results showed that even low dose of 177Lu‑RDC inhibited LNCaP tumor growth, with inhibition increasing as the dose rose; high dose achieved nearly 100% tumor growth suppression (Figure 6), demonstrating a clear dose–response relationship.
Figure 6. Efficacy evaluation of 177Lu‑RDC in the LNCaP model.
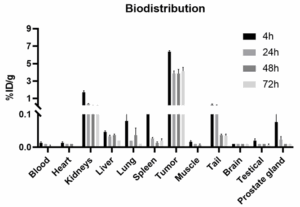
We also assessed the biodistribution of 177Lu‑RDC in LNCaP tumor‑bearing mice at multiple time points (Figure 7). The results showed high accumulation of 177Lu‑RDC in tumor tissue, further confirming the targeting advantage of RDCs.
Figure 7. Biodistribution of 177Lu‑RDC in LNCaP tumor‑bearing mice
We established a dual‑tumor model by implanting 22RV1 cells on the left flank and LNCaP cells on the right flank of male BALB/c nude mice. When tumors reached ~300 mm3, mice were treated with 177Lu‑RDC per the study protocol. The results showed that 177Lu‑RDC produced significant tumor growth inhibition in LNCaP tumors but had no inhibitory effect on the PSMA‑low/medium 22RV1 tumors (Figure 8), consistent with the PET imaging findings (Figure 5C, D).
Figure 8. Efficacy evaluation of 177Lu‑RDC in the dual‑tumor model
WuXi Biology is empowering RDC drug discovery and development
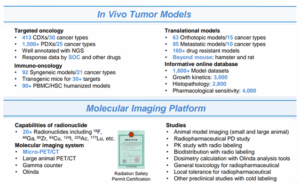
WuXi Biology focuses on preclinical research of RDCs, offering a one‑stop solution of RDCs that includes radiolabeling, in vivo imaging in large and small animals, preclinical efficacy studies, and pharmacokinetics. These services help partners accelerate RDC drug discovery and development (Figure 9).
Figure 9. Tumor Models and Molecular Imaging Platform at WuXi Biology
References
- Tran, H.H., Yamaguchi, A. & Manning, H.C. Radiotheranostic landscape: A review of clinical and preclinical development. Eur J Nucl Med Mol Imaging 52, 2685–2709 (2025).
- https://clinicaltrials.gov/
- Bidkar AP, Zerefa L, Yadav S, VanBrocklin HF, Flavell RR. Actinium-225 targeted alpha particle therapy for prostate cancer. Theranostics. 2024 May 11;14(7):2969-2992. doi: 10.7150/thno.96403. PMID: 38773983; PMCID: PMC11103494.
- Li, Z.; Ruan, Q.; Jiang, Y.; Wang, Q.; Yin, G.; Feng, J.; Zhang, J. Current status and perspectives of novel radiopharmaceuticals with heterologous dual-targeted functions: 2013–2023. J. Med. Chem. 2024, 67, 21644– 21670.
Related Content
Building on the momentum of AACR 2026, WuXi AppTec was honored to host an outstanding group of regional industry leaders...
VIEW RESOURCEBuilding on the momentum of AACR 2026, WuXi AppTec was honored to host an exceptional group of regional industry leaders...
VIEW RESOURCE