PD-1 Blockade Protein in 4th Generation Armored CAR-T Cells Enhances Cytotoxicity Effect with in vitro Re-challenge System
Chimeric antigen receptor (CAR)-T cells are genetically engineered T cells that express a surface receptor to recognize tumor-associated antigens (TAA). CAR-T cell therapy have demonstrated remarkable success with hematological malignancies. However, limited headway has been made towards solid tumor, due to various challenges, including recognition of tumor-specific antigen trafficking and penetration, localization and survival within an immunosuppressive tumor microenvironment (TME) [1].
Overexpression of immunosuppressive cytokines and/or proteins, such as TGF-β and PD-L1, respectively, downregulates cytotoxic CD8+ T cells, reducing the efficacy of CAR-T cell therapy. Immune checkpoint inhibitors (ICIs), such as anti-PD-1/PD-L1 and anti-CTLA-4 antibody have gathered immense attention due to their efficacy across multiple solid malignancies [2]. As such, a combination of CAR-T and ICIs may prove to be a viable strategy to overcome an unfavorable TME.
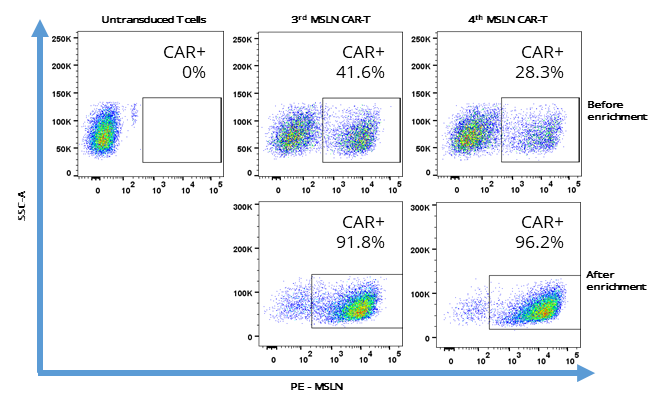
Here, we have established a 4th generation, armored CAR-T coupled with a PD-1 blockade protein, to enhance the anti-exhaustion effect of CAR-T cells. Our results indicate that the addition of a PD-1 blockade protein reduces the amount of available PD-1 on the surface in both CAR-positive and -negative T cells, reduces exhaustion of CAR-T cells, and prolong cytotoxic killing effect of CAR-T cells in a 192 h in vitro re-challenge assay compared to 3rd generation CAR-T.

PD-1 blockade protein in 4th generation armored CAR-T cells enhances cytotoxicity effect with in vitro re-challenge system
Related Content
Small cell lung cancer (SCLC) remains one of the most aggressive and difficult-to-treat cancers, highlighting the ongoing need for new...
VIEW RESOURCEFrom hematologic malignancies to solid tumors Introduction: Amid a surge of advancements in immunotherapy, T cell engager (TCE) therapy has...
VIEW RESOURCE
