mRNA Drugs: Progress, Production, and Prospects

Introduction:
RNA-based therapies are a frontier in biomedicine, with the potential to fundamentally change treatments for a wide range of diseases. The COVID-19 pandemic has significantly accelerated the industrialization and clinical translation of mRNA technology. In addition, research on mRNA therapeutics for other infectious diseases, cancer treatment, protein replacement therapy, and in vivo cell therapy is increasingly a major focus. This review summarizes the research progress, manufacturing processes, challenges, and future outlook of mRNA therapeutics, and further shares the R&D strategies for mRNA drugs.
The Rise of mRNA Therapeutics
In 1990, Wolff demonstrated that injecting in vitro–transcribed mRNA into mouse skeletal muscle cells resulted in expression of the desired protein, thereby introducing the concept of mRNA therapeutics [1]. With breakthroughs in mRNA modification and delivery technologies, mRNA drugs have rapidly advanced. The COVID-19 pandemic further accelerated clinical validation and translation of mRNA therapeutics. Moderna’s mRNA-1273 and BioNTech/Pfizer’s BNT162b2, owing to their safety and efficacy, became the first mRNA products to receive FDA authorization [2].
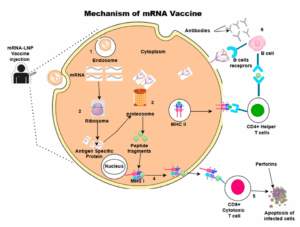
mRNA vaccines are taken up by cells and translated in the cytoplasm into antigen proteins, which are then processed and presented on MHC molecules to activate CD8+ cytotoxic T cells and CD4+ helper T cells, ultimately inducing both cellular and humoral immune responses to eliminate pathogens (Figure 1) [3]. Research on mRNA vaccines against influenza, rabies, Zika virus, human metapneumovirus (hMPV), respiratory syncytial virus (RSV), herpes simplex virus (HSV), human immunodeficiency virus (HIV), and others is accelerating. On May 31, 2024, Moderna’s RSV mRNA vaccine mRESVIA (mRNA-1345) received FDA approval for the prevention of RSV-related lower respiratory tract disease in adults aged 60 and older. This marks Moderna’s second approved mRNA vaccine and signals continued maturation of clinical translation for mRNA therapeutics [2].
Figure 1. Mechanism of action of mRNA vaccines [2]
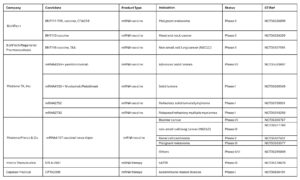
In addition to prophylactic vaccines, mRNA therapeutics are rapidly advancing in therapeutic cancer vaccines, protein replacement, and in vivo cell therapies (Table 1).
Table 1. Ongoing clinical trials related to mRNA therapeutics [2]
Advantages of mRNA Therapeutics
Over the past decade, breakthroughs in mRNA therapeutics have conferred distinct advantages in vaccine development and protein-replacement therapies. Compared with conventional biologics (e.g., inactivated vaccines, subunit vaccines, DNA vaccines, and therapeutic antibodies), mRNA therapies offer:
- Low risk of insertional mutagenesis: mRNA is non-integrating and is naturally degraded by physiological processes; its half-life and immunogenicity can be tuned via chemical modifications and delivery strategies [3].
- Rapid design and flexible encoding: mRNA sequences can be easily swapped to encode different proteins, shortening development timelines. As a minimal genetic carrier, mRNA can be dosed repeatedly and, with suitable delivery systems, is efficiently taken up and translated in the cytoplasm [4].
- Scalable, platform-based production: high-yield in vitro transcription and an increasingly mature supporting industry enable fast, low-cost, platformed, large-scale manufacturing.
Manufacturing process of mRNA therapeutics
Plasmid DNA preparation and in vitro transcription
Production begins with a plasmid DNA (pDNA) template containing a DNA-dependent RNA polymerase promoter and the target mRNA sequence. The pDNA is linearized by restriction digestion and used as a template for in vitro transcription with enzymes such as T7 RNA polymerase.
A mature mRNA has two key structural features:
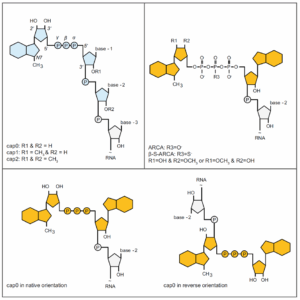
- 5′ cap (Figure 2): protects mRNA from exonuclease degradation and binds translation initiation factors to promote ribosome loading and translation. The cap can be formed enzymatically (e.g., guanylyltransferase to make Cap0, followed by methyltransferase to produce Cap1) or introduced as a cap analogue during transcription to simplify the process.
- 3′ poly(A) tail: composed of tens to hundreds of adenosine residues, it protects mRNA from degradation and binds poly(A)-binding protein (PABP) to facilitate ribosome recognition and translation initiation. The poly(A) tail can be encoded in the pDNA template or added enzymatically after transcription.
Figure 2. Schematic of different mRNA cap structures [4]
Sequence optimization and nucleotide modification
mRNA translation is influenced by multiple factors, notably codon adaptation, mRNA secondary structure (minimum free energy, MFE), and GC content, all of which significantly affect mRNA stability and translational efficiency. For example, using species- or even cell-specific codon choices and avoiding rare codons can markedly improve translation.
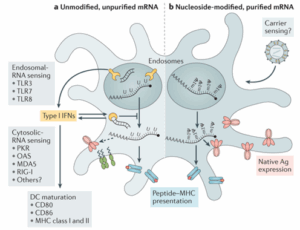
Nucleotide modifications are another key factor. Unmodified mRNA robustly activates innate immunity, reducing intracellular stability and translation efficiency (Figure 3). Incorporating modified nucleotides can substantially dampen innate immune activation—for instance, pseudouridine or N1-methylpseudouridine incorporation significantly suppresses interferon-induced activation of PKR (double-stranded RNA–dependent protein kinase) and related intracellular immune signaling, thereby reducing immune responses [8,9].
Figure 3. Innate immune sensing of mRNA [10]
Targeted delivery of mRNA therapeutics
A central challenge for mRNA therapy is the efficient delivery of mRNA molecules to specific tissues or cells. Lipid nanoparticles (LNPs), the dominant mRNA delivery vehicles, protect mRNA from nuclease degradation and immune activation, facilitate cellular uptake, and promote endosomal escape into the cytoplasm. Compared with other delivery systems such as liposomes or polymer carriers, LNPs offer higher encapsulation efficiency and lower carrier-associated toxicity. Advances in ionizable lipids and microfluidic manufacturing have enabled stable, scalable production of high‑encapsulation‑efficiency mRNA formulations. Notably, targeted delivery of mRNA provides a valuable tool for organ- or cell‑selective protein expression, opening new avenues for precision therapeutics [11].
Case Study
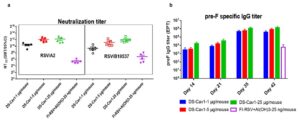
WuXi Biology successfully formulated and systematically evaluated an RSV mRNA vaccine. Mice received a prime on day 0 and a boost on day 21, and immunogenicity was compared experimentally. Results showed that, compared with a licensed aluminum‑adjuvanted inactivated RSV vaccine, the mRNA vaccine induced significantly higher neutralizing antibody titers with a clear dose‑dependent effect (Figure 4a). A pronounced dose response was also observed for antibodies binding to the RSV pre‑F antigen (Figure 4b).
Figure 4. Neutralizing antibody titers and pre‑F antigen‑binding antibody titers in mice
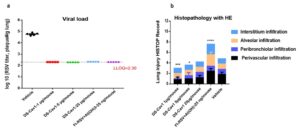
After immunizations on days 0 and 21, mice were challenged with RSV A2 on day 42. On day 47, lung viral loads and pulmonary pathology were assessed. All vaccine groups showed lung viral loads near the detection limit (Figure 5a), while the adjuvanted inactivated vaccine caused marked inflammatory infiltration and tissue damage compared with the mRNA vaccine (Figure 5b).
Figure 5. Lung residual viral loads and H&E-stained lung histopathology in mice
On day 49, T‑cell activation was assessed by ELISpot and flow cytometry. The mRNA vaccine markedly enhanced T‑cell responses, with a significant increase in the proportion of IFN‑γ–positive cells in vaccinated mice (Figure 6).
Figure 6. ELISpot analysis of T lymphocytes in mice
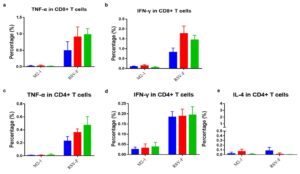
Flow cytometry results showed higher TNF-α and IFN-γ secretion from CD8+ T cells than from CD4+ T cells. Within CD4+ T cells, TNF-α and IFN-γ levels were significantly higher than IL-4 (Figure 7), indicating the mRNA vaccine induces a strong Th1‑biased immune response.
Figure 7. Flow cytometric analysis of CD8+ and CD4+ T lymphocytes in mice
Conclusion
mRNA technology represents a revolutionary advance in biomedicine, showing great potential across infectious diseases, cancer therapy, protein replacement, and cell-based treatments. It also holds promise for personalized medicine by enabling tailored therapies that maximize efficacy while minimizing adverse effects.
WuXi Biology has established an integrated mRNA drug discovery platform. We support partners with services including mRNA sequence design and optimization; mRNA synthesis and formulation; targeted delivery (organ, tissue, and specific cell populations); quality assessment; in vitro expression and functional validation; in vivo efficacy and early safety evaluation. These capabilities comprehensively empower mRNA drug development across indications such as infectious disease, oncology, protein replacement, and in vivo cell therapies (e.g., in vivo CAR).
References:
[1] Wolff, J A et al. “Direct gene transfer into mouse muscle in vivo.” Science. 247,4949 Pt 1 (1990): 1465-8.
[2] Oloruntimehin, Sola et al. “mRNA Vaccine Technology Beyond COVID-19.” Vaccines. 13,6 601. 31 May. 2025.
[3] Perenkov, Alexey D et al. “In Vitro Transcribed RNA-Based Platform Vaccines: Past, Present, and Future.” Vaccines. 11,10 1600. 16 Oct. 2023.
[4] Chlake, Thomas et al. “mRNA as novel technology for passive immunotherapy.” Cellular and molecular life sciences. 76,2 (2019): 301-328.
[5] ltkamp, Silke et al. “Modification of antigen-encoding RNA increases stability, translational efficacy, and T-cell stimulatory capacity of dendritic cells.” Blood. 108,13 (2006): 4009-17.
[6] Rudzien-Nogalska, Ewa et al. “Synthesis of anti-reverse cap analogs (ARCAs) and their applications in mRNA translation and stability.” Methods in enzymology. 431 (2007): 203-27.
[7] Enderson, Jordana M et al. “Cap 1 Messenger RNA Synthesis with Co-transcriptional CleanCap® Analog by In Vitro Transcription.” Current protocols. 1,2 (2021): e39.
[8] Schwartz, Schraga et al. “Transcriptome-wide mapping reveals widespread dynamic-regulated pseudo uridylation of ncRNA and mRNA.” Cell. 159,1 (2014): 148-162.
[9] Kowalczyk, Aleksandra et al. “Self-adjuvanted mRNA vaccines induce local innate immune responses that lead to a potent and boostable adaptive immunity.” Vaccine. 34,33 (2016): 3882-93.
[10] Ardi, Norbert et al. “mRNA vaccines – a new era in vaccinology.” Nature reviews. Drug discovery. 17,4 (2018): 261-279.
[11] Nuphar Veiga et al. “Cell specific delivery of modified mRNA expressing therapeutic proteins to leukocytes” Nat Communication. 2018 Oct 29;9(1):4493.
Related Content
DNA-encoded libraries have become widely used in drug discovery, and several approaches for linking chemical compounds to DNA have been...
VIEW RESOURCETargeted protein degradation (TPD) technology has become one of the most promising methods to remove specific disease-related proteins using cellular...
VIEW RESOURCE