Expanded Role of PDX Models in Anticancer Drug Development
Expanded Role of PDX Models in Anticancer Drug Development
Patient-derived xenograft (PDX) models are established by implanting surgically resected patient tumor tissues into immunodeficient mice and have become a pivotal tool in preclinical cancer research due to high clinical-relevance. These models faithfully preserve the original tumor’s molecular, histopathological and pharmacological characteristics, reflecting the heterogeneity and diversity of human tumors. Importantly, these biological characteristics in PDX models remain stable through serial passaging in mice, offering significant improvements over standard cell-line derived xenograft (CDX) models. Owing to these properties, PDX models have found extensive application in anticancer drug screening, biomarker discovery and precision medicine.
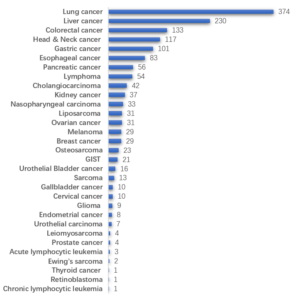
WuXi Biology has pioneered the development of over 1500 PDX models across 30 cancer types, encompassing both prevalent and rare indications for solid and hematological malignancies. These models are derived from primary, recurrent or metastasis patients with or without clinical treatments (Figure 1). Each model is comprehensively characterized in terms of molecular, tissue pathology and in vivo pharmacology.
Clinical information, growth dynamics, histopathology, drug sensitivity data, and molecular profiles for each PDX model are integrated into our comprehensive tumor model online database. With its powerful search engine, users can quickly apply filters to identify models that meet their research needs under multi-condition settings.
Figure 1: Overview of PDX model database of WuXi AppTec
While PDX models stand out as one of the most ideal preclinical in vivo models, they do have limitations, including modeling complexity, lengthy timelines, high costs, and the requisite use of immunodeficient animals. These factors can impede large-scale screening and immuno-oncology drug development. To overcome these limitations, we have leveraged our extensive PDX model database to expand PDX applications at the in vitro, ex vivo and in vivo levels by developing PDX-derived cell lines and organoids, ex vivo PDX assays, drug-resistant PDX models, orthotopic and metastatic PDX models, humanized PDX models, and the PDX “clinical trial” capability. These advancements broaden the application of PDX models in anticancer drug development, thereby optimizing the cost, speed, and quality of preclinical drug development.
PDX-Derived Cell Lines and Organoids
PDX-derived cell lines (PDC) represent in vitro tumor models that are developed by extracting tumor cells from freshly harvested PDX tumor tissues and cultivating them ex vivo for a short duration. Unlike commercially available human cell lines, this model maintains the basic molecular characteristics and biological phenotypes of the original PDX, thereby offering greater clinical relevance. Currently, we have developed multiple PDX-derived cell lines covering 14 types of tumors, including lung cancer, pancreatic cancer, liver cancer, colorectal cancer, glioblastoma, and lymphoma.
PDX-derived organoids (PDXO) represent another form of PDX-derived in vitro tumor models. Fresh PDX tissues are dissected and cultured in 3D conditions within a basement membrane matrix enriched with specific growth factors. This process simulates the tumor matrix environment, forming a tumor organoid model. PDXOs closely retain the molecular, tissue pathology, and pharmacological characteristics of their source PDX tissues, reproducing the heterogeneity within tumors and between individuals. We have developed multiple PDX-derived organoid models, representing various types of tumors such as colorectal cancer, head and neck cancer, lung cancer, pancreatic cancer, and gastric cancer.
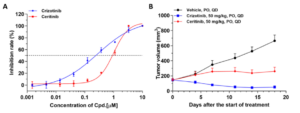
Both PDCs and PDXOs are well annotated by whole-exome and RNA-sequencing and pharmacological validation, ensuring reproduction of the molecular features and pharmacological phenotypes of the corresponding PDX models. These models can be used in the development of targeted oncology and immuno-oncology drugs, supporting in vitro high-throughput and high-content drug screening and evaluation with stronger clinical relevance. They can also guide the selection of appropriate PDX models for in vivo studies (Figure 2).
Figure 2: Study of ALK inhibitor efficacy in a lung cancer PDC carrying ALK gene fusion and its corresponding PDX model
Ex Vivo PDX Assays
Ex vivo PDX assays involve the isolation and transfer of primary cancer cells from fresh PDX tissues to a suitable in vitro environment for prompt drug activity testing. Isolated primary cancer cells fully retain the features of the original PDX models and can be utilized to test candidate compounds before in vivo PDX studies are initiated, significantly saving research time and cost. To support this approach, we have developed PDX-based ex vio experimental systems such as the tumor chemosensitivity assay (TCA).
TCA involves digesting freshly separated PDX tumor tissues into single cells, obtaining tumor cells by density gradient centrifugation, and then using the CellTiter-Glo® (CTG) method for antitumor drug screening and evaluation. This method is commonly used to assess drug cytotoxicity and is one of the routine methods for cytotoxic drug screening and evaluation (Figure 3).
Figure 3: Flowchart of the tumor chemo-sensitivity assay
Drug-Resistant PDX Models
Drug resistance largely limits clinical benefits of cancer therapies. As such, the development of drug-resistant models to investigate resistance mechanisms and identify mechanism-based strategies to overcome resistance has become a crucial step in preclinical drug discovery and development. At WuXi Biology, we have developed clinical drug-resistant PDX models and induced drug-resistant PDX models. By combining in vivo pharmacology and molecular analysis, we aim to facilitate the exploration of resistance mechanisms and the development of next-generation anticancer drugs or combination therapies designed to overcome resistance.
Orthotopic and Metastatic PDX Models
Figure 4: Study of ROS1 inhibitor efficacy in a lung cancer brain metastasis PDC-luc model carrying ROS1 gene fusion
To better simulate clinical disease progression and the tumor microenvironment based on PDX models and thereby improve clinical translation in anti-cancer drug development, we have labeled cell lines derived from PDX models with luciferase and used them to construct in vivo orthotopic and metastatic models, providing reliable guidance for drug development, especially against advanced malignancies (Figure 4).
Humanized PDX Models
With the development of cancer immunotherapy, humanized tumor models engrafted with human immune cells and bearing human tumor xenografts have become a key tool for preclinical research. As a representative of these models, humanized PDX models combine the clinical features of original patient tumors with characteristics of human immune cells, which can support immuno-oncology drug discovery with higher clinical translation. We have successfully developed several PDX models reconstituted with human PBMCs, NKs or HSCs. These models cover five types of cancers, including lung cancer, head and neck cancer, melanoma, liposarcoma, and kidney cancer, and are used for assessing immuno-oncology drugs that target various immune cells, such as T cells, NK cells and myeloid cells (Figure 5).
Figure 5: Testing the efficacy of PD-1 antibody in EGFR wild-type, mutant and overexpressing PDX mouse models with humanized immune systems
PDX “Clinical Trials”
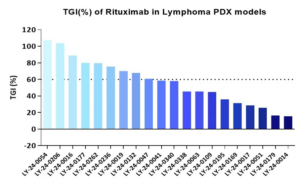
“PDX clinical trials (PCTs)” simulate the design of human clinical trials to assess population-level drug responses in a set of PDX cohorts, which represent an accurate and efficient approach for assessing the clinical potential of some therapeutic modalities. By integrative pharmacogenomic analysis, it enables insight into inter-patient response heterogeneity and helps to identify potential clinically relevant resistance mechanisms and discover or validate informative biomarkers providing opportunities to develop strategies that are likely to be translated in the clinic (Figures 6 and 7).
Figure 6: Translational research of PDX clinical trials
Figure 7: PDX clinical trials for treating diffuse large B-cell lymphoma with rituximab
WuXi AppTec | Tumor Models:
Related Content
AACR 2026 Posters | Sneak Peek The Annual Meeting of the American Association for Cancer Research (AACR) will be held...
VIEW RESOURCEAt AACR 2026, scientists from our WuXi Biology platform will present a collection of the company’s latest oncology research findings...
VIEW RESOURCE