Engineered cell line panel complemented with in vivo models to enable next-generation target therapy discovery in cancer
AACR Annual meeting 2021
Authors: Zhixiang Zhang1, Feifei Fan2, Xiaohe Shi2, Xiangyang Zuo2, Qingyang Gu2, Qunsheng Ji1. 1 WuXi AppTec, Shanghai, China; 2 WuXi AppTec, Jiangsu, China Corresponding Author: Qunsheng Ji. Email: ji_qunsheng@wuxiapptec.com
Background
Many cancer types are driven by specific gene aberrances, like gene fusions, mutations and copy number changes. It has led to the successful application of a number of target therapeutics, of which kinase inhibitors represent a good example. However, acquired resistance is almost inevitable at the end of any target therapy treatment, which makes it urgent a need to discover new-generation therapeutics. There are strong evidences suggesting acquired resistance should be largely attributed to certain secondary mutations, such as EGFRT790M, RETV804L and BTKC481S.
At discovery stage it is critical to generate preclinical cancer models harboring these specific gene aberrances both in vitro and in vivo. Biopsy from patient with clinical resistance could be an optional starting point. But it is much more straightforward and efficient if these well-studied gene aberrance could be constructed and engineered into host cell lines directly. The transformed cell line can further be inoculated into host mice to make in vivo test feasible.
Methods
A series of cancer driver gene aberrances were constructed, including activating gene fusions or mutations, and the corresponding secondary mutations which confer resistance to existing target agents. These constructs were transfected into BaF3 cell line, respectively. After rounds of in vitro selection, the stable BaF3 cell lines harboring these driver gene aberrances were subjected into in vitro activity test of different generations of target therapeutics. Meanwhile, these cell lines were also implanted into immuno-deficient animals subcutaneously. When tumor volumes reached When average tumor volume reached 100-200 mm3, the tumor bearing mice were randomized and administered with different generations of target therapeutics for in vivo efficacy evaluation.
Results
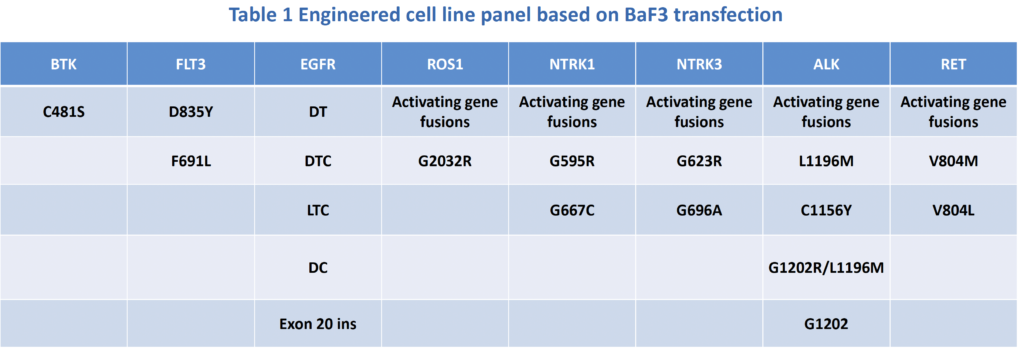
A panel of engineered BaF3 cell lines have been established, focusing at driver genes in lung cancer, lymphoma, and many other indications. For each driver gene target, there are wild type or activating gene fusion lines, which do not contain resistant mutations. Such gene overexpression or aberrance transformed BaF3 cell lines to be independent of IL-3, which are always sensitive to early generation of target therapeutics. Besides, the well-recognized drug resistant mutations are also constructed and transfected into BaF3 lines. These cell lines can be valuable for new generation of target therapeutics aiming to conquer the emerging acquired drug resistance.
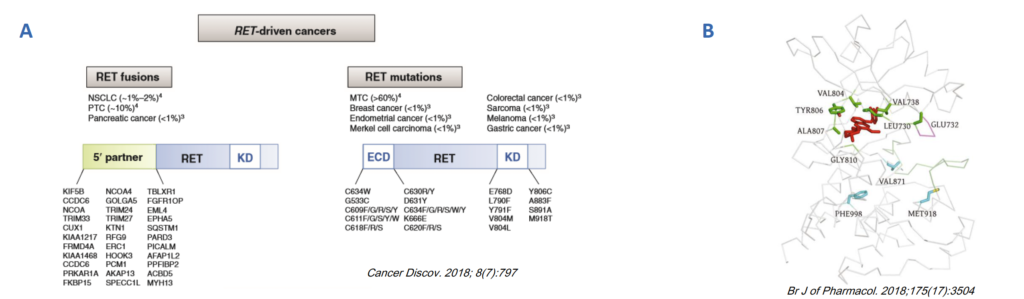
Fig 1 Activating gene aberrances and acquired resistant mutations (RET as an example)
A. Aberrant activation of the RET gene have been associated with multiple tumorigenesis. RET fusions are somatic juxtapositions of 5′ sequences from other genes with 3′ RET sequences encoding tyrosine kinase that lead to constitutively active cytosolic chimeric proteins. B. Some RET mutations are reported to confer tumor resistance to kinase inhibitors, many of which are located in the Gly-rich loop (L730, E732 and V738), the gate-keeper residue (V804) or the hinge strand (Y806, A807 andG810).
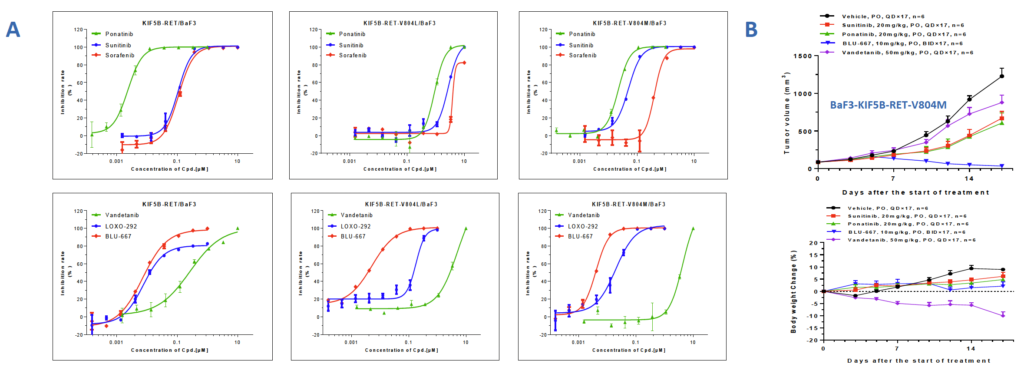
Fig 2 Engineered cell line validation in vitro and in vivo (RET as an example)
A. The stable BaF3 cell line harboring KIF5B-RET, KIF5B-RET-V804L or KIF5B-RET-V804M was subjected into in vitro test of six kinase inhibitors with RET inhibitory activity, including Ponatinib, Sunitinib, Sorafenib, Vandetanib, LOXO-292 and BLU- 667. As selective next generation of RET inhibitor, BLU-667 and LOXO-292 demonstrated superior in vitro activities to other multiple kinase inhibitors when suppressing the two V804 mutated cell line growths. B. In KIF5B-RET-V804M transformed BaF3 tumor model, BLU-667 also demonstrated much improved efficacy compared with other three agents.
Conclusion
In summary, our ever-expanding panel of engineered cell lines complemented with in vivo models represents valuable assets to empower the discovery of next-generation target therapeutics against cancer.
References
- Subbiah V, Gainor JF, Rahal R, Brubaker JD, Kim JL, Maynard M, Hu W, Cao Q, Sheets MP, Wilson D, Wilson KJ, DiPietro L, Fleming P, Palmer M, Hu MI, Wirth L, Brose MS, Ou SI, Taylor M, Garralda E, Miller S,Wolf B, Lengauer C, Guzi T, Evans EK. Precision Targeted Therapy with BLU-667 for RET-Driven Cancers. Cancer Discov. 2018 Jul;8(7):836-849. doi: 10.1158/2159-8290.CD-18-0338. Epub 2018 Apr 15. PMID: 29657135.
- Liu X, Shen T, Mooers BHM, Hilberg F, Wu J. Drug resistance profiles of mutations in the RET kinase domain. Br J Pharmacol. 2018 Sep;175(17):3504-3515. doi: 10.1111/bph.14395. Epub 2018 Jul 19. PMID: 29908090; PMCID: PMC6086982.
- Iams WT, Lovly CM. Stop fRETting the Target: Next-Generation RET Inhibitors Have Arrived. Cancer Discov. 2018 Jul;8(7):797-799. doi: 10.1158/2159-8290.CD-18-0489. PMID: 29967074; PMCID: PMC6345252.

2021 AACR engineered cell line panel_1118_final to AACR_F
Related Content
High-dimensional (HD) flow cytometry allows researchers to perform deep phenotyping of immune cells at the single cell level with increased...
VIEW RESOURCEWe offer an extensive panel of cutting-edge immune profiling and biomarker discovery services to support: Target discovery and validation Drug...
VIEW RESOURCE
